
I am often asked the question: “Do I need a defoamer or an antifoam?” The answer is neither yes nor no, for there is a difference between them.
Many customers contact me because they want to solve their ‘foaming’ problem – something they cannot control. The problem is typically heavy surface foam which is generated in various locations in the pulp or papermaking process. Wire tray, wire silo, drains, effluent treatment plant, size press, coating color kitchen, brown stock washers, and blow tank are a few examples. Surface foam can lead to various problems. We need to focus underneath the surface of the liquid where the foam starts as an air bubble.
Why do air bubbles and surface foam occur?
To form a stable foam, we need to meet the following criteria:
- A liquid (water is a major component of the pulp and paper making process)
- An impurity in the liquid (there are many chemicals and surfactants found naturally in wood which are released during the pulping process; paper makers use many additives in the wet end to give the paper different functional properties)
- Mechanical energy (the process of making pulp and paper is very dynamic)
Pulp and papermaking systems contain all these three criteria; therefore, foaming is a common occurrence.
Air bubble is generated in the liquid phase by mechanical energy and occasionally by chemical or biological reaction. Air in the liquid phase is known as dissolved gas, entrained air or free air and the size of the air bubble dictates whether it remains in suspension or rises to the surface of the liquid. The impurities present in the liquid phase (such as surfactants) dictate the stability of the air bubble in both the liquid phase and at the surface of the liquid.
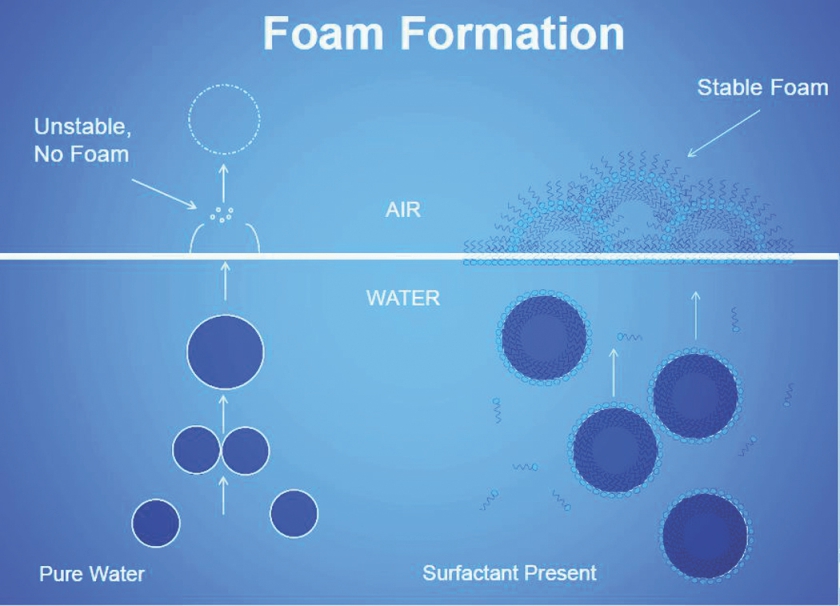
Air and foam stabilizers (surfactants) are common in the liquid phase: In the pulping process, the natural wood lignin is converted into soaps at high temperature and pH; they foam excessively. In paper making, additives such as dry strength, wet strength, sizing and starch are a few examples of chemicals that stabilize air and foam.
If the stabilized air bubble remains in the liquid phase, it contributes to various negative aspects of the production process such as:
Paper
- Poor formation resulting in reduced sheet strength, reduced production due to sheet breaks and sheet defects such as pinholes and light spots
- Reduced pump efficiency resulting in reduced production
- Reduced drainage resulting in reduced production and higher steam consumption
Pulp
- Poor washing efficiency resulting in dirtier pulp
- Lower liquor solids resulting in higher energy costs at the evaporator
- Higher soda loss resulting in more alkali to purchase
- Decreased VAT dilution resulting in slower speed / drainage
- Increased dirt count resulting in dirty pulp
- Increased bleaching costs
- Knock on effects to paper machine and ETP
If the stabilized air bubble rises to the liquid surface it will not burst and will remain until physically or chemically destroyed. Surface foam has some of the following negative aspects: –
- Water level in chests difficult to control
- Deposition on the walls of chests resulting in microbiological problems
- Dried foam on the walls breaking off causing sheet defects and breaks.
- Housekeeping / safety / appearance (foam in drains, rivers etc.)
- Loss of additives
- ETP foaming problems create environmental risks
All of the above add $$$$$ to the production cost, which an effective antifoam / defoamer can significantly reduce.
How do we control the air in liquid and surface foam?
The air in the liquid phase and surface foam can be controlled by mechanical and chemical treatment.
Mechanical
- The use of water spray/shower bars directly over the surface foam will physically knock down the foam
- Deculators are vacuum systems designed into the engineering of the mill; the liquid phase is put under reduced pressure at which point the air can be easily removed
Chemical
- A defoamer reduces or destroys existing surface foam
- An antifoam reduces the air content in the liquid phase and prevents surface foam from stabilizing; if this can be achieved, there is less need for a defoamer
- An effective foam control agent should combine both effects to some extent
How do we choose the correct foam control agent?
Like all chemical applications, there are some basic rules we should follow.
“I recommend using both defoamer and antifoam for mills that can benefit from reduced entrained air and surface foam.”
For defoamers, we target heavy surface foam. We simply need to test different chemical types in order to see which one effectively destabilizes the surface foam. The most effective defoamer chemical type will likely be a surfactant, but opposite to the surfactant types which stabilize the foam. After we have chosen the most suitable defoamer to use, we need to select the best addition point. It is important to select a positon with good mixing to ensure even distribution of the defoamer. Because the defoamer is fast acting it can be added just before or at the point of heavy foaming.

For antifoams, we target a reduction in the liquid phase entrained air. Product selection is easier. The most effective antifoams used in the paper making wet end are typically fatty alcohol dispersions and stock temperature dictate the product of choice.
I recommend using both defoamer and antifoam for mills that can benefit from reduced entrained air and surface foam. Pulp mill foam control agents are a combination of defoamer and antifoam; they are typically silicone-based emulsions or concentrates and are designed for use at high temperature and high pH.
Finally, the pulp and paper making process is dynamic and never stable, which means that defoamer and antifoam performance can also change. It is therefore advisable to regularly survey the products used in order to ensure optimum performance.
– The article was written by Richard West, Product Manager, Amazon Papyrus Chemicals



