As paper mills move toward highly closed-loop systems, the focus extends beyond how much water can be reduced to how these systems behave under constraint, and what defines their practical limits.

Over the past decade, the pulp and paper industry has made significant strides in reducing freshwater consumption through recycling and closed-loop systems. Leading mills operating with advanced process integration and high levels of internal water circulation have demonstrated that substantial reductions are technically achievable. The question, however, has changed now: not how far consumption can fall, but where it must hold.
From Reduction to Stabilization
At the forefront of this transition are large, globally integrated producers such as Suzano and APP, where water management systems have evolved into disciplined operational regimes. These mills report that freshwater consumption has begun to stabilize, reflecting a balance between reuse, system constraints, and process requirements. This stabilization is not indicative of stagnation but rather signals that these systems are operating within defined technical boundaries.
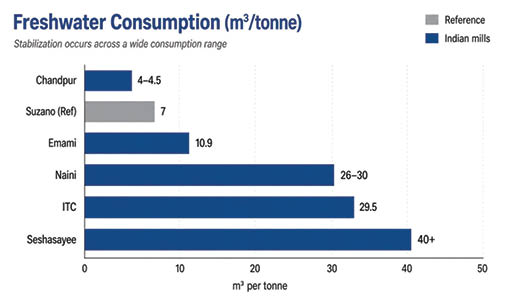
A similar trend is visible across Indian paper mills, albeit across a wider range of operating conditions. At ITC’s Bhadrachalam unit, specific freshwater consumption has stabilized at around 29.5 m³/tonne. Emami Paper Mills has sustained a level of 10.9 m³/tonne, while Chandpur Paper operates at approximately 4–4.5 m³/tonne.
Despite the variation in absolute values, the common thread is stabilization. As Naini Papers observes, this reflects a “closed-loop equilibrium,” where internal recycling loops are balanced against unavoidable system losses. In practical terms, this means that further reductions are no longer driven by incremental improvements alone but are constrained by the inherent dynamics of the system itself.
This change marks an important transition in how water management is understood within the industry. The earlier phase focused on how far freshwater consumption could be reduced through technological adoption and process improvements. The current phase is increasingly concerned with how these highly recycled systems behave under sustained operation, and what factors define their practical limits. As ITC notes, “…the achievable limit for further water reduction is defined by a balance of technical feasibility, product quality requirements, system reliability, and economic considerations.”
Watch: Top Paper Companies 2023
The Plateau Is Not a Problem, It Is a Signal
The stabilization of freshwater consumption across mills is often interpreted as a slowdown in progress. The stabilization of freshwater consumption across mills reflects a more fundamental change, i.e., the system has reached an operating balance.
As Naini Papers notes, “Stability indicates that the mill has reached a ‘closed-loop equilibrium’,” where internal recycling loops are balanced against unavoidable system losses. At this stage, water becomes part of a dynamic system where accumulation, dilution, and removal are continuously balanced.
Several mills report that once this equilibrium is reached, further reduction becomes difficult without altering system conditions. At ITC’s Bhadrachalam unit, freshwater consumption has remained steady over recent years, maintained through careful water balance management and continuous monitoring of process circuits. Emami Paper Mills similarly observes that it is operating close to its practical minimum under current conditions, with further reductions constrained by the need to maintain product quality and system reliability.
This plateau reflects the limits of the current operating configuration rather than the theoretical minimum. Beyond this point, further reductions are no longer a matter of incremental optimization but would require structural changes in how water is treated, distributed, or used within the system.
Even among global producers operating at lower freshwater consumption levels, a similar pattern is evident. Once high system closure is achieved, the challenge shifts from reducing water intake to managing its internal consequences.
In this context, the plateau marks the transition from efficiency-driven reduction to constraint-driven operation. Understanding what defines this equilibrium, and what disrupts it, becomes essential to identifying how far water reuse can realistically be extended.
The Cost of Reuse
As mills push toward greater water reuse and tighter system closure, operational challenges shift inward, now driven by the accumulation of contaminants within recirculating water streams.
Across mills, a key issue is the buildup of dissolved and suspended substances, such as fibers, fines, dissolved organics, and salts, that are not fully removed through conventional treatment. At ITC’s Bhadrachalam unit, this leads to deposit formation requiring periodic “boil-out program” for paper machines to maintain system cleanliness. Satia Industries similarly reports wet-end instability, foaming, and reduced drainage efficiency under higher reuse conditions.
At the chemical level, Naini Papers notes that dissolved impurities, or “anionic trash,” interfere with retention aids and strength additives, affecting paper quality and machine performance. Microbial activity in recycled water further contributes to slime formation and runnability issues.
These challenges are not isolated to individual mills but are inherent to the physics and chemistry of closed-loop systems. From a technology perspective, Voith notes that elevated COD levels in recirculated water can stimulate biological activity, resulting in acidification, scaling, and operational instability.
From a process chemistry standpoint, the impact of such accumulation is equally significant. Kemira highlights that rising TDS and conductivity levels often necessitate higher chemical dosing, which in turn places additional demands on water usage for dilution and distribution. In recycled and brown-grade production, excessive water reuse can breed odorous compounds, like volatile fatty acids, hydrogen sulfide, quietly undermining product quality and market acceptability.
Taken together, these observations mark a conspicuous change: water reuse is not simply a question of how much can be recycled, but how its quality evolves within the system, and how that interacts with process chemistry, equipment, and product outcomes.
Closed-loop systems behave as dynamic environments rather than static infrastructure. The challenge is not just maximizing reuse, but managing its internal consequences while preserving stability and product integrity.
Why Limits Exist
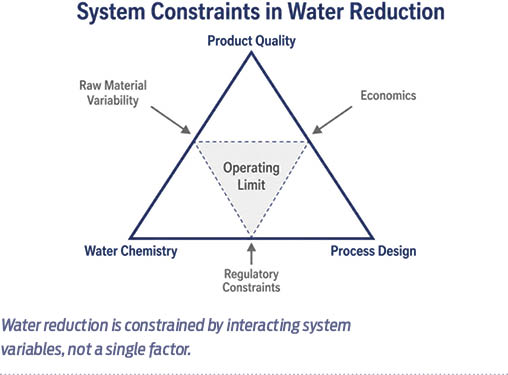
While operational challenges reveal how water behaves under high reuse, the more critical question is why further reduction becomes constrained. Across mills, this limit is not defined by a single factor, but by the interaction of process requirements, water chemistry, and product specifications.
Product quality is a primary constraint. In papermaking, water is not merely a utility; it is an active component that directly influences fiber bonding, brightness, colour, and surface properties. At Emami Paper Mills, water quality directly affects brightness and shade, while ITC notes that key operations, such as chemical preparation and pulp processing, require water of specific quality, limiting reuse, limiting the extent to which recycled water can be used without affecting output quality.
Raw material variability further complicates this. Mills processing agro-residues or recycled fiber streams introduce a higher load of non-process elements (NPEs) such as silica, extractives, inks, and dissolved organics into the system. As Satia Industries points out, these elements accumulate within closed loops and affect wet-end chemistry, equipment reliability, and microbial control, ultimately constraining further reduction in freshwater usage. As Naini Papers notes, the increasing concentrations of dissolved substances can lead to issues such as brightness reversion and corrosion.
At a system level, these limitations reflect the chemistry of recirculated water. Voith notes that once such contaminants reach saturation, they disrupt the functional chemistry of the paper machine, leading to quality defects and increased instability. In parallel, Kemira emphasizes that elevated TDS and conductivity levels not only affect chemical dosing requirements but also necessitate additional water input for dilution, thereby counteracting further reductions in freshwater consumption.
Even in highly optimized systems, these constraints define a practical boundary. As Suzano indicates, the limits of water reuse are governed by physicochemical thresholds within the process, beyond which stability and product quality cannot be maintained without significant changes to system design or treatment capability.
As Emami succinctly puts it, “The technical and operational factors that define the practical limit for reduction in water consumption are: high purity water requirements for sensitive grades, equipment and system design constraints, and build-up of dissolved contaminants and microbiological load.”
These factors show that the limits of water reduction are not simply technological. They are intrinsic to how papermaking systems operate. Product requirements, raw material characteristics, and water chemistry define a boundary that cannot be bypassed through incremental improvements.
The Need to Purge
The question now is not whether water can be recycled, but how long it can remain within the system before its quality begins to compromise operations. Purging thus emerges as a necessary requirement of closed-loop systems.
Across mills, there is clear recognition that contaminants cannot be indefinitely retained within recirculating water streams. Without controlled removal, they reach levels that disrupt stability, damage equipment, and affect product quality.
In this context, purging functions as a regulatory mechanism within the system. At Seshasayee Paper and Boards, “Purging is the ‘safety valve’ of a closed-loop system. To maintain a steady state, we periodically remove water to mitigate the buildup of COD, BOD, and color carryover… This controlled purge essentially defines our ‘freshwater floor’—the absolute minimum intake required to dilute contaminants and ensure sustained performance.”
This perspective is echoed across other mills, though expressed through different operational practices. At Naini Papers, purging is framed as a necessary step to control the buildup of NPEs such as potassium and silica, particularly in agro-based systems where such constituents are inherently higher. Satia Industries similarly notes that periodic or continuous purging is required to manage dissolved solids, organics, and microbial load in closed loops. Even in systems where direct purging is minimized, as in the case of ITC, controlled discharges in specific circuits are maintained to keep TDS and conductivity within acceptable operating limits.
From a system design perspective, this requirement is intrinsic. Voith explains that purging prevents contaminant buildup that would otherwise “poison the process chemistry,” and defines a physical limit: “the absolute minimum freshwater requirement is determined by the ‘evaporation threshold’… at this stage, the only fresh water added is the amount required to replace what is lost to the atmosphere during the drying process and for chemical preparation.”
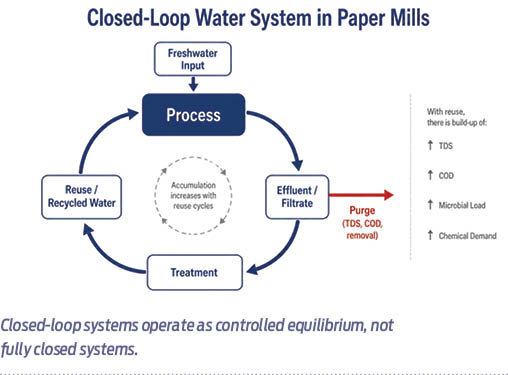
These insights show that closed-loop systems are not fully closed; they operate as controlled equilibrium systems. Purging is therefore not inefficiency, but a necessary condition for maintaining balance.
In practical terms, it is the purge rate, rather than the reuse rate, that defines how far freshwater consumption can be reduced. Beyond a point, the need for dilution establishes a lower bound that cannot be eliminated without redesigning the system.
Water Use by Requirement
As closed-loop systems approach their operational limits, water within the mill ceases to be a uniformly distributed utility and becomes a resource that must be actively managed and allocated. The focus shifts from maximizing reuse in aggregate to determining where and how different qualities of water can be used without compromising process stability or product performance.
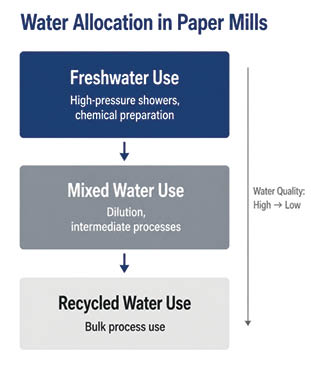
Across mills, this takes the form of a “fit-for-purpose” approach. Freshwater is treated as a high-value input and reserved for critical operations such as high-pressure showers, chemical preparation, and key process stages where quality requirements are stringent.
Suzano articulates this approach as a structured allocation strategy, where freshwater is intentionally directed toward high-sensitivity applications, while recycled streams are deployed across less critical sections. This enables the mill to maintain operational reliability while maximizing overall reuse within the system.
A similar logic is evident across Indian mills. At Naini Papers, freshwater is reserved for what are described as “critical path” areas, such as chemical dilution and high-pressure showers, where water purity directly influences the effectiveness of additives and equipment performance. Emami Paper Mills follows a water “quality tiering approach”, allocating freshwater to critical sections like sealing systems and dilution circuits, while relying on recycled streams for the bulk of process requirements. ITC similarly bases allocation decisions on the tolerance of different process areas to parameters such as TSS, TDS, and chemical residues. Even at low consumption levels, Chandpur Paper applies the same principle, using freshwater strategically while substituting recycled water wherever feasible.
As Chandpur Paper explains, “Freshwater is used in areas where higher water quality is critical, while treated and recycled machine effluent is used in other process stages, allowing us to minimize freshwater dependency without compromising product quality.”
In this framework, water systems operate as interconnected networks rather than linear flows. Allocation decisions are influenced by product mix, raw material variability, and system conditions, determining how close a mill can operate to its minimum freshwater requirement.
Control Through Measurement
As water systems become increasingly closed and complex, stability can no longer be maintained through fixed practices alone. Mills rely on continuous monitoring and responsive control to manage fluctuations in water quality and process conditions. Monitoring thus becomes the basis for operational decision-making.
Across mills, key parameters such as TDS, COD, pH, conductivity, and microbial load are tracked to ensure water quality remains within acceptable limits for reuse. At ITC, additional parameters such as fiber content, colour, and Optical Brightening Agent (OBA) levels are monitored to ensure compatibility with product requirements and process conditions. Deviations in these indicators directly influence decisions on whether water can be reused, requires further treatment, or must be partially discharged.
The growing complexity of these decisions has led to the adoption of real-time monitoring systems and digital tools. At Emami, online flow meters and tracking systems enable proactive adjustments, while APP relies on continuous monitoring of parameters such as pH, TSS, TDS, and conductivity to maintain process stability.
From a systems perspective, this data-driven approach is becoming essential in managing high-reuse environments. Voith notes that digital platforms enable real-time tracking of parameters such as COD, flow rates, and chemical dosing, allowing mills to anticipate and correct deviations within the water cycle.
At the same time, process chemistry considerations remain closely linked to monitoring practices. Kemira emphasizes that maintaining high levels of water reuse while ensuring product quality depends on monitoring and controlling chemical parameters. Without such control, fluctuations in water composition can lead to increased chemical consumption, instability in wet-end chemistry, and inconsistent product characteristics.
These developments reflect a new operational philosophy. As systems approach their limits, stability is achieved by managing variability within the system, with monitoring acting as the feedback mechanism that enables operation close to minimum freshwater levels.
The ability to measure, interpret, and respond to changing water conditions thus becomes a defining capability for mills operating under high system closure.
Extending the Limits
If current operating conditions define the present limits of water reduction, further progress will depend on how those conditions evolve. Incremental optimization will yield only marginal gains; meaningful reductions will require structural changes in water use, treatment, and control.
For global producers such as Suzano, this transition is already framed in terms of system-level improvement rather than incremental efficiency. Further reductions in freshwater consumption are linked to advances in process integration, treatment capability, and operational discipline, rather than isolated upgrades. The emphasis is on strengthening internal loops while maintaining the physicochemical balance required for stable operation.
A similar direction is emerging among Indian mills, though shaped by different operational realities. Satia Industries identifies advanced water treatment technologies, such as membrane filtration, ion exchange, and ZLD systems, as necessary to push beyond current reuse levels, particularly in managing dissolved solids and organics within closed loops. Naini Papers points toward emerging approaches such as advanced oxidation processes (AOPs) and digital twin modelling, which can help predict system load and optimize chemical dosing in real time, thereby enabling higher levels of reuse without destabilizing the process.
However, these pathways are constrained by economics. ITC notes that while advanced treatment can enable further reuse, the capital and operating costs must be justified against marginal gains at already low consumption levels. This introduces a critical dimension to the discussion: the next stage of water reduction is not only a technical challenge but also an economic one.
From a system design perspective, there is also a growing shift toward source-level intervention. Seshasayee Paper and Boards emphasizes the need to move beyond end-of-pipe treatment toward addressing contaminants at their point of origin within the process. By targeting streams, such as colored filtrates and high-TDS flows earlier in the cycle, mills can reduce the burden on downstream treatment systems and recover larger volumes of usable water.
Technology providers reinforce this direction. Voith advocates for tiered treatment architectures that combine biological processes with advanced filtration to enable safe operation at lower freshwater consumption levels. At the same time, digital systems are expected to play a larger role in predicting system behavior and optimizing process conditions dynamically. Kemira similarly points to the importance of improved chemical strategies and monitoring frameworks in managing water chemistry under increasingly constrained conditions.
Despite these developments, the fundamental constraint remains unchanged: reductions must be achieved without compromising product quality or process reliability. As Chandpur Paper notes, “Any future reductions in freshwater consumption would likely depend on advancements in treatment technologies and further improvements in system efficiency, while ensuring that product quality standards are maintained.”
This underscores the nature of the transition ahead. The next stage will require a rethinking of system architecture: integrating treatment, process design, and digital control in a way that allows mills to operate closer to their theoretical limits while maintaining stability and economic viability.
Also Read: ITC PSPD Aligns With CPCB Standards, Backs Economically Viable & Scalable Water Recycling, Reusing Solutions
From Circularity to System Management
The trajectory of water management in the pulp and paper industry has moved through a clear sequence. What began as a focus on reduction has evolved into a phase where reuse is embedded in operations. Today, the industry faces a more complex challenge: operating effectively within systems that are already highly closed.
Across both global and Indian contexts, freshwater consumption has declined significantly but is now stabilizing. This stabilization reflects the convergence of process requirements, water chemistry, and system dynamics, defining a practical operating boundary.
Within this boundary, mills must manage contaminant accumulation, chemical interactions, and operational variability while maintaining product quality. Purging defines system limits, allocation optimizes usage, and monitoring enables control. Together, these elements form the basis of modern water management.
The implication is clear: the industry has largely learned how to reuse water. The emerging challenge is learning how to operate with it.


